Medical technology
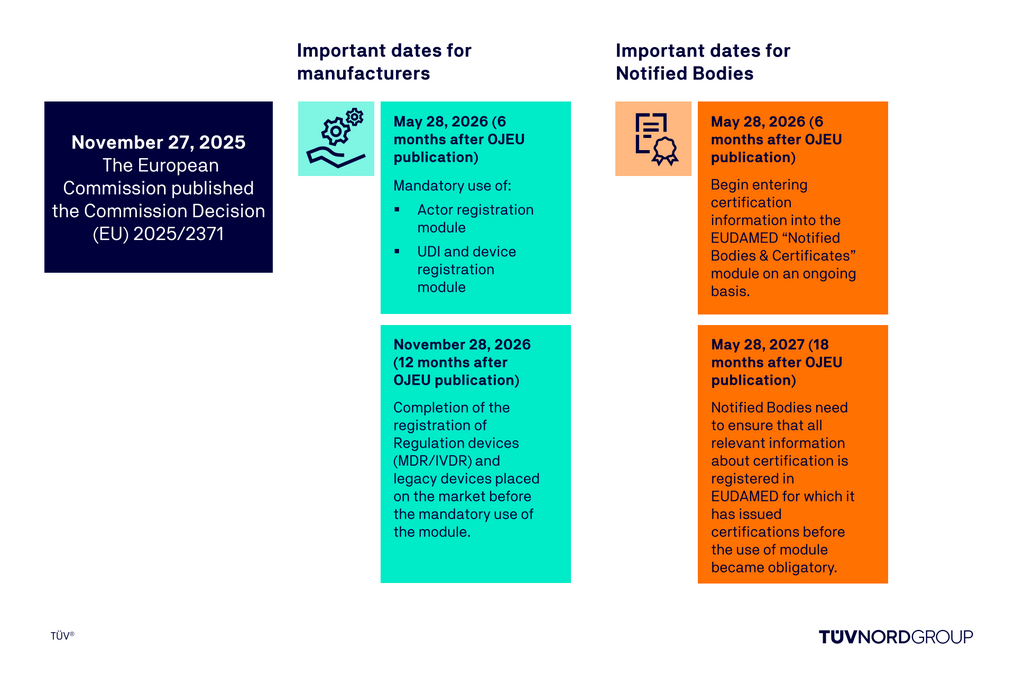
EUDAMED: Mandatory use of first four modules from May 28, 2026
March 24, 2026
The European Commission is adopting important regulatory requirements in relation to EUDAMED: The first four EUDAMED modules are now fully functional. Our overview outlines the changes and applicable deadlines, helping manufacturers prepare for the new EUDAMED obligations in a timely manner.
Contact us now