Assured quality of cosmetic ingredients
Buyers of cosmetic products are ever more sensitive to the ingredients and processes used in their manufacture. In order to meet the rising expectations of customers in the sector, European Federation for Cosmetic Ingredients has developed the Good Manufacturing Practice Standard (EFfCI GMP).




Benefits of certification
Certification to the EFfCI-GMP Standard by TÜV NORD CERT as an independent certification body offers:
-
recognised evidence that suitable facilities, equipment and production controls are in place in order to produce cosmetic ingredients of assured quality
-
Clear and convincing arguments which you can use to access new markets and customer groups
• recognised evidence that suitable facilities, equipment and production controls are in place in order to produce cosmetic ingredients of assured quality
• Clear and convincing arguments which you can use to access new markets and customer groups
Your EFfCI Certification Process
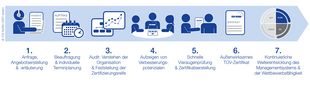
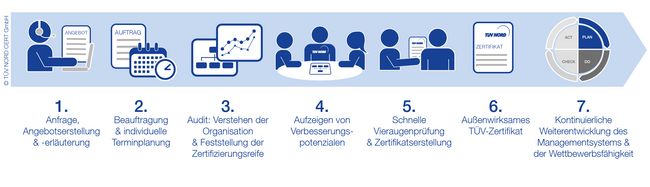
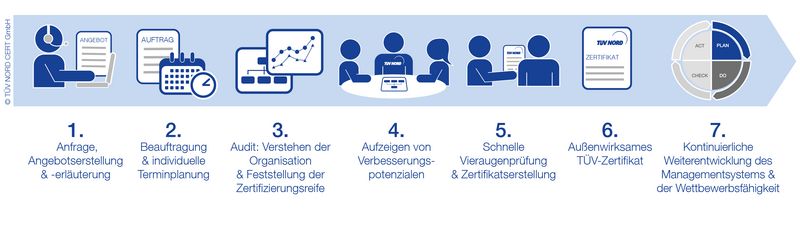
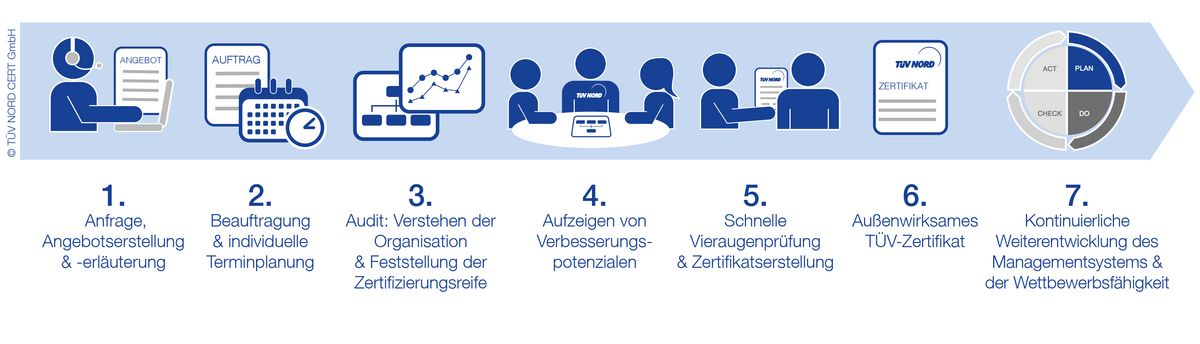
FAQ
The abbreviation EFfCI stands for The European Federation for Cosmetic Ingredients. The EFfCI is a European industry association that has represented the common interests of more than 100 manufacturers of cosmetic ingredients in Europe since 2000.
The EFfCI-GMP certificate is valid for three years. Annual surveillance audits and a recertification audit after three years are carried out to maintain the certificate.
EFfCI certification is aimed at all raw material supplying companies and sales organizations in the cosmetics industry.
The EFfCI GMP Guidelines are based on the structure of ISO 9001 and the IPEC (International Pharmaceutical Excipients Council) GMP Guide. The EFfCI GMP certification is a voluntary extension of the ISO 9001 certification.
Our service includes:
-
Certification audit
-
Nonconformity management
-
Evaluation of corrective actions and drafting of reports
-
Issue of certificate and transfer of a copy to the EFfCI, with validity of 3 years subject to annual surveillance audits.
Your international sales contact in Germany
TÜV NORD CERT GmbH
Food & Agriculture
Tel.: +49 (0)160 888 3919
Food-Certification@tuv-nord.com
International Sales Contact by Country
CCPB Italy
Mrs. Elisabetty Szulin
eszulin@tuv-nord.com
Tel: +39 (0)3701300804